Gradient Spin Echoes for Selective Excitation
Shaped excitation pulses can replace the non-selective hard pulses typically used in a one-pulse measurement to achieve
selective excitation. Another method of achieving selective excitation is the gradient spin echo using a selective 180° pulse. This technique is demonstrated in the figure below.
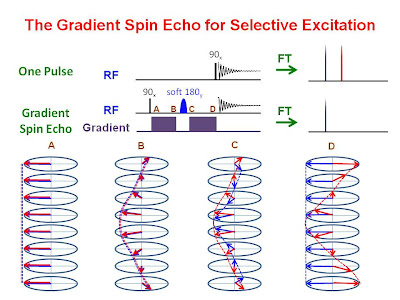
A non-selective hard 90°x pulse is first given followed by a pair of identical pulsed field gradients sandwiching a soft selective 180° pulse about the y axis. The hard 90° pulse rotates all spin vectors onto the -y axis. During the first gradient pulse the spin vectors
dephase and
evolve according to their offset frequencies. The soft 180°y pulse flips a single resonance 180° about the y axis leaving all other resonances untouched. During the second gradient pulse, the "selected" resonance is rephased and its
offset frequency evolution is refocused. The unselected resonances dephase more and continue to evolve according to their offset frequencies. The receiver is then turned on to collect the FID of the "selected" resonance, all others are dephased and therefore suppressed. This is demonstrated in the figure below which shows 1H NMR spectra for a mixture of methylence chloride and acetone.

The bottom trace shows a standard one-pulse measurement. The middle and top traces show results from a selective gradient spin echo measurement with the selective 180° pulse set for methylene chloride and acetone, respectively.



Source:
University of Ottawa NMR Facility Blog